�U�ی����������Ҷ�������PET����ͨ�^����Ӛ��D����߃rֵ���WƷ��չ�F����đ��Ý�����Ȼ�������F���ʵĴ����������R�����ڱ��о��У��о��ˆT�״����þ����OӋ�� PtW/MCM-48 ������ͨ�^�����{�ش������е�����λ�c�����F�ˏU PET һ偷��x���ԼӚ��Ƃ䌦�������ᣨp-TA�����a�ʸ��_53.4%��ͬ�r�a36.4%�Č����ױ���PX�����C���о��������䃞���Ĵ�����Դ�� Pt �{���w���c WOx ��N֮�g�ąfͬ���ã��̓r WOx ��N������ Pt �ķ�ɢ�ԣ��� Pt �Mһ������ WOx ��;ۺϑB���u���}��PtW1.5/MCM-48 �߂��m����λ�c������Ч�{�� p-TA �Ľ������������^�ȼӚ��D���� PX�������挍 PET �U���D����չ�F�����õ��m���ԡ�LCA �� TEA �����Mһ���@��ԓ���g�đ���ǰ����
���ͻ��W���ռ��g���J����һ�N���F�U��PET�ĸ�Ч�D����ֵ���õ����뷽����������֪�����������ᣨp-TA����һ�N��Ҫ�Ļ���ԭ�ϣ��V�����ڸй���ϡ�������������ЙC�ϳɡ�Ŀǰ��p-TA�Ĺ��I���a����ه����ʯ���X�������Č����ױ���PX����ԭ�ϣ�ͨ�^������������h����⒴��Ă��yʯ��;���������O�丯�g���ء�p-TA�a�ʵ͡������o�����յȼ��ֆ��}�����֮�£����ͻ��W���ռ��g�ڂ��y�Ļ��W���չ�ˇ�Ļ��A�ϣ�ԭλ������ۺ�Ć��w����PET���ӽY���б��h�c�Ȼ��Ĺ�ܗЧ�������䲻���Q�Ӛ䷴���O�����y�����F�����Q�仯�a��ĸ߮a����Ȼ��һ���ش����𡣿˷��@Щ�����ϵK��Ҫ�_�l�߶�ᘌ��Ժ��x���ԵĴ�ϵ�y�����д������m������OӋ�ʬF����ͬ�����ď��s�Ժ��y�ȡ�
�������c
1�����о�����һ�����x���ԼӚ���ԣ����F�U PET ���x���������Q�Ӛ������Ƃ���ֵ���WƷ p-TA���m���ڶ�N��ʽ�� PET �U�ϡ����������u�r��LCA���ͼ��g�����u�r��TEA���Y��������ԓ�������H�@���p���˂��yʯ����ˇ�����ĭh����Ⱦ��ͬ�r�߂䃞Խ�Ľ��������ԡ�
2�����о����þ����OӋ���p��PtW/MCM-48������չ�F��Խ�Ĵ����ܣ�ʹ p-TA �� PX �����ʷքe�_�� 53.4% �� 36.4%�����������^�ߵķ����ԣ����x����ѭ�h���á�
3����N�����ֶκ�DFTӋ��Y����ͬ������PtW/MCM-48�����ă�Խ����Դ��Pt NPs��WOx��N֮�g�ąfͬ����ã������� Pt/W �����x������m�ȵ��е�����λ�c�����Ծ��ʿ������g�w�������ͽ������Ķ����Mp-TA���γɣ�ͬ�r�����^�Ӛ�����PX��
4�����о��_�l��һ�N��Ч�Ľ���/���p���ܴ�������������ˏU PET �x���ԼӚ��^�����ʻ��h�ͱ��h�IJ��֚仯�C�������U���Ϛ仯�����¼��g���_�l������Ҫ�����rֵ��
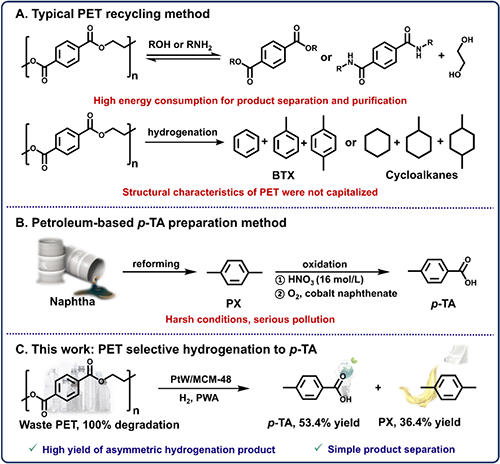
Figure 1. Schematic diagrams of conventional PET recycling methods, petroleum-based p-TA preparation, and the new method for p-TA production via selective hydrogenation of PET in this work.
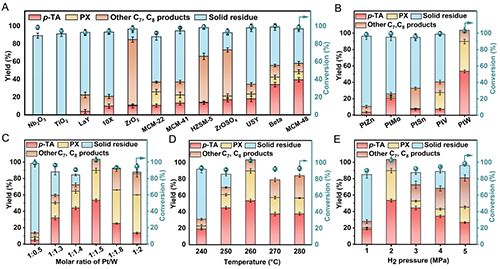
Figure 2. Optimization of reaction conditions for targeted hydrogenation of waste PET to p-TA and PX over Pt-based catalysts. (A) Effect of different supports; (B) effect of second metal sites of bimetallic Pt-M/MCM-48 (n(Pt): n(M) = 1:1.5); (C) effect of Pt/W molar ratio in PtW/MCM-48 catalyst; (D) effect of reaction temperature; (E) effect of H2 pressure. General reaction conditions: 0.05 g PET, 3 mL of H2O, 12 h, 700 rpm, 0.05 g catalyst with 5.0 wt% Pt loading, pH=2, 260 ��C, 2 MPa H2.
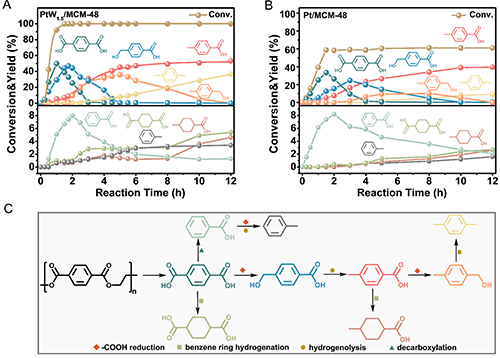
Figure 3. (A, B) Reaction product distributions on the targeted hydrogenation of waste PET over (A) PtW1.5/MCM-48 and (B) Pt/MCM-48 catalysts as a function of time. Reaction conditions: 0.05 g PET, 3 mL of H2O, 260 ��C, 2 MPa H2, 12 h, 700 rpm, 0.05 g catalyst, pH=2 (with PWA). Specially, for aromatic liquid products, the yields were calculated by (moles of aromatic ring in product)/ (moles of aromatic ring in feedstock). For raw PET materials, the conversion was calculated by (moles of all aromatic liquid products)/ (moles of aromatic ring in feedstock). (C) Possible reaction pathway for the targeted hydrogenation of waste PET over PtW1.5/MCM-48 catalyst.
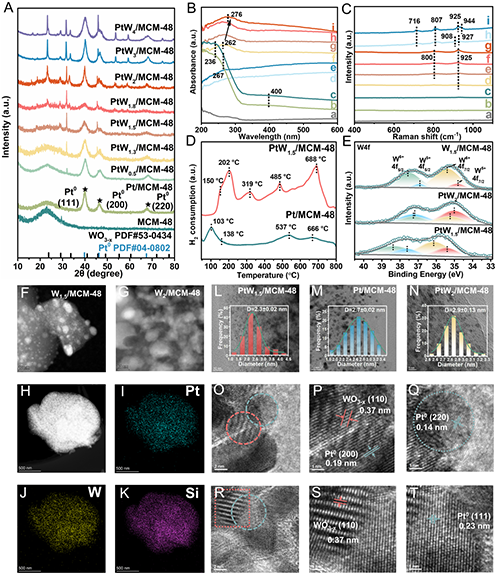
Figure 4. (A) XRD patterns of MCM-48, Pt/MCM-48 catalyst, and PtW/MCM-48 catalysts with different Pt/W molar ratios. (B) UV-vis spectra of (a) MCM-48, (b) W1.5/MCM-48, (c) W2/MCM-48, (d) Pt/MCM-48, (e) PtW0.5/MCM-48, (f) PtW1.3/MCM-48, (g) PtW1.5/MCM-48, (h) PtW1.8/MCM-48, (i) PtW2/MCM-48. (C) Raman spectra of (a) MCM-48, (b) Pt/MCM-48, (c) PtW0.5/MCM-48, (d) PtW1.3/MCM-48, (e) PtW1.5/MCM-48, (f) PtW1.8/MCM-48, (g) PtW2/MCM-48, (h) W1.5/MCM-48, (i) W2/MCM-48. (D) H2-TPR profiles of the Pt/MCM-48 and PtW1.5/MCM-48 catalysts. (E) XPS spectra of W4f in the W1.5/MCM-48, PtW2/MCM-48 and PtW1.5/MCM-48 catalysts. (F, G) HAADF-STEM images of (F) W1.5/MCM-48 and (G) W2/MCM-48 samples. (H-K) EDX spectroscopy mapping profiles of PtW1.5/MCM-48 catalyst. (L-N) HR-TEM images and particle size distributions of (L) PtW1.5/MCM-48, (M) Pt/MCM-48, (N) PtW2/MCM-48 catalysts. (O-T) HR-TEM images of the PtW1.5/MCM-48 catalyst.
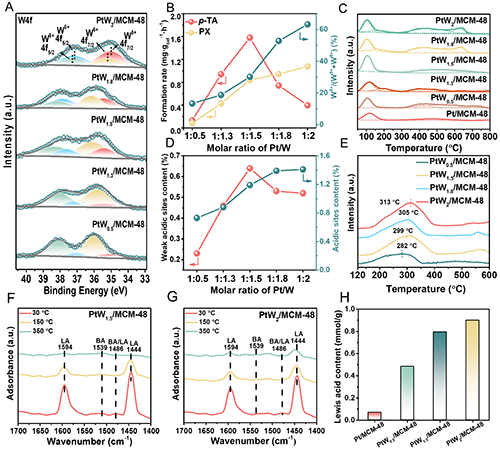
Figure 5. (A) XPS spectras of W4f in the PtW/MCM-48 catalysts with different PtW molar ratios. (B) Correlation between the molar ratio of Pt/W of PtW/MCM-48 catalysts and the formation rate of p-TA and PX, along with the W5+/(W5++W6+) ratio on the catalysts surface. (C) NH3-TPD profiles of the PtW/MCM-48 catalysts. (D) Relationship between Pt/W molar ratio in PtW/MCM-48 catalysts and the content of weak and total acidic sites. (E) Acetic acid TPD-MS results for PtW/MCM-48 catalysts with different PtW molar ratios. (F, G) The IR spectra of adsorbed pyridine on (F) PtW1.5/MCM-48 and (G) PtW2/MCM-48 catalysts. (H) Br?nsted and Lewis acid sites contents and ratios of Pt/MCM-48 and PtW/MCM-48 catalysts.
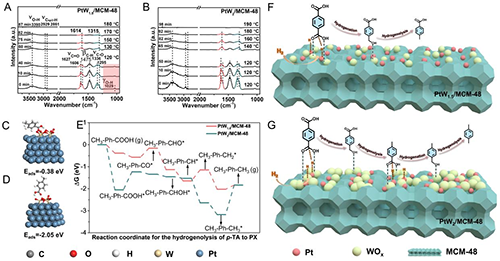
Figure 6. (A, B) DRIFTS images of acetic acid adsorption on (A) PtW1.5/MCM-48 and (B) PtW2/MCM-48 catalysts. (C, D) The intermediate CH3-Ph-COOH* on the (C) W3O7/Pt(111) and (D) W4O7/Pt(111) model. (E) Reaction energies of the hydrogenolysis of p-TA on W3O7/Pt(111) and W4O7/Pt(111) surfaces. (F, G) Schematic diagram of selective hydrogenation of waste PET to p-TA and PX over (C) PtW1.5/MCM-48 and (D) PtW2/MCM-48 catalysts.
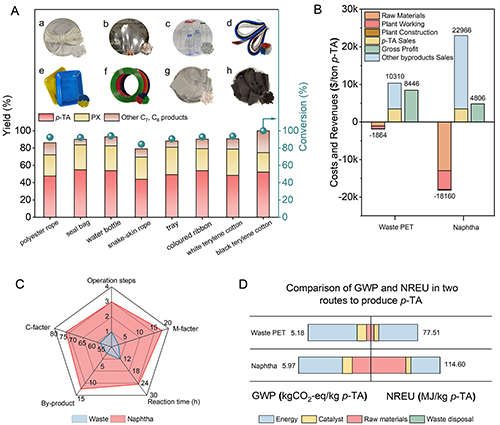
Figure 7. (A) Targeted hydrogenation of real waste PET plastics (a) polyester rope, (b) seal bag, (c) water bottle, (d) snake-skin rope, (e) tray, (f) coloured ribbon, (g)white terylene cotton, (h)black terylene cotton to p-TA and PX over PtW1.5/MCM-48 catalyst. General reaction conditions: 0.05 g real PET plastics, 3 mL of H2O, 260 ��C, 2 MPa H2, 700 rpm, 12 h, 0.05 g PtW1.5/MCM-48, pH=2 (with PWA). (B) Comparison of the revenues and costs of waste PET and naphtha to p-TA (100,000 tons p-TA production per year). (C) Comparison of the viabilities of the production of p-TA from waste PET and naphtha. ��M-facter�� referred as Material consumption (ton/ton p-TA) and ��C-facter�� referred as Catalyst cost ($/ton p-TA). (D) Comparison of non-renewable energy use (NREU) and global warming potential (GWP) in production of 1 kg p-TA from waste PET and naphtha.
ԭ����Ϣ��
��һ���ߣ���Խ��ë�ܷf
ͨӍ���ߣ�÷����
ͨӍ��λ���㽭��W�h���c�YԴ�WԺ
Փ��DOI��10.1021/jacs.5c01209
https://pubs.acs.org/doi/10.1021/jacs.5c01209
ͨӍ���ߺ���

÷�������㽭��W�h���c�YԴ�WԺ������Ӌ�����о��T����ʿ��������Ӣ���ʼҌW��ţ�D���H�W�ߡ��L�������������|/�U���ϵ��ЙC���w�U����ĸ�ֵ�����ã����@�P�I���W�I�ľ�����c�{�أ��_�lԭ�ӽ������D��·���c�Gɫ���wϵ���Ԍ��F���s����|�ЙC�̏U�wϵ�ĸ��x�����YԴ���ա���Sci. Adv., J. Am. Chem. Soc., Angew. Chem. Int. Ed. �ș����ڿ��ϰl��SCIՓ��50��ƪ���ڙ����Ұl������13�������և�����Ȼ�ƌW�����㽭ʡ���c�аlӋ�����Ŀ������Science Bulletin/���ƌWͨ�� ������ί��The Innovation�ڿ����꾎ί��Carbon & Hydrogen���꾎ί��
��Ҫ�о������U����/�����|�ȹ̏U�YԴ�����ЙC�̏U���h�����ܲ��ϣ���Դ�̏U�fͬ�������g�c�b�䡣
E-mail: [email protected]
�̎���퓣�https://person.zju.edu.cn/qingqingmei
