蔣林波 電合成與電催化 2025年09月12日10:05 中國香港
第一作者:蔣林波,陳興寶
通訊作者:木士春教授
通訊單位:武漢理工大學(xué)
研究背景:
深入理解水氧化反應(yīng)即析氧反應(yīng)(OER)催化機(jī)制對(duì)于設(shè)計(jì)高性能催化劑及提升整體電解水能量轉(zhuǎn)換效率具有重要意義。目前,相較于典型的單位點(diǎn)吸附演化機(jī)制(AEM)與晶格氧介導(dǎo)機(jī)制(LOM),通過鄰近金屬活性中心的吸附氧物種直接耦合的雙位點(diǎn)氧化路徑機(jī)制(OPM)成為單位點(diǎn)機(jī)制研究的延伸,促進(jìn)了整體催化反應(yīng)動(dòng)力學(xué)與穩(wěn)定性,近年來受到人們廣泛關(guān)注。然而,相較于Ir/Ru基貴金屬OER催化劑,非貴金屬(No/Co/Fe)位點(diǎn)的低本征活性與復(fù)雜的表面重構(gòu)行為嚴(yán)重阻礙了其OPM機(jī)制研究與結(jié)構(gòu)開發(fā)。此外,對(duì)于Ni/Fe基過渡金屬催化劑,異質(zhì)雙位點(diǎn)通常具有不同的中間體吸附傾向性(*O/*OH),不利于其直接的兩步去質(zhì)子化后的同步*O-O*耦合。因此,利用Ni-Fe鄰近雙位點(diǎn)的不一致中間體演變行為,并合理調(diào)控其局域空間配置與原子排布,遵循Ni-OH-O-Fe差異性氧化路徑機(jī)制(DOPM),有望觸發(fā)雙位點(diǎn)路徑以極大提升其催化活性與穩(wěn)定性。本文中,鑒于Ni/Fe位點(diǎn)分別有利于*OH與*O中間體吸附,通過氧陰離子(PO43-)改性觸發(fā)了Ni-Fe雙活性中心的不同步去質(zhì)子化過程,實(shí)現(xiàn)能壘更低的*OH-O*耦合過程,即差異性氧化路徑(DOPM),從而用于高效穩(wěn)定的電解水產(chǎn)氫。
主要?jiǎng)?chuàng)新點(diǎn)和結(jié)論
1. 理論分析與計(jì)算研究:DFT計(jì)算表明,氧陰離子(PO43-)浸出過程改善了羥基氧化物(R-PO4-NiCoFeOOH)的空間配置、能帶結(jié)構(gòu)與電子分布,并優(yōu)化了Ni-Fe/O的配位構(gòu)型、原子間距及合適的*OH/*O吸附能,有利于實(shí)現(xiàn)Ni-Fe高價(jià)鄰近位點(diǎn)的 DOPM路徑。自由能計(jì)算揭示了R-PO4-NiCoFeOOH具有更高的Ni/Fe位點(diǎn)本征活性,在PO43-浸出位點(diǎn)附近觸發(fā)了Ni-Fe雙位點(diǎn)*OH-O*耦合的DOPM路徑,促進(jìn)了催化活性與穩(wěn)定性。
2. 結(jié)構(gòu)表征:相關(guān)結(jié)構(gòu)表征揭示了PO43-融入了NiFe LDH晶格,并誘導(dǎo)了OER極化過程中的深度原位重構(gòu),實(shí)現(xiàn)了高無定形、均勻分布且高價(jià)態(tài)的羥基氧化物活性物質(zhì)的形成,同時(shí)避免了Fe物種的偏析與聚集,有利于鄰近位點(diǎn)的直接催化反應(yīng)并提升長期穩(wěn)定性。PO4物種改性促進(jìn)了表面原子重排與電子重分布,實(shí)現(xiàn)了有著更飽和配位及更短雙金屬距離的Ni-O-Fe橋連物種,有利于觸發(fā)雙位點(diǎn)協(xié)同的DOPM路徑。
3. 機(jī)制研究:原位質(zhì)譜與光譜等證明了在PO43-離子浸出與氧物種填充誘導(dǎo)的深度重構(gòu)下,R-PO4-NiCoFeOOH催化劑中AEM與重構(gòu)誘導(dǎo)的浸出位點(diǎn)附近局域DOPM機(jī)制共存,形成了異質(zhì)Ni-*OH-O*-Fe橋氧中間體,與理論預(yù)測(cè)分析一致。
4. 電化學(xué)性能:R-PO4-NiCoFeOOH展現(xiàn)了優(yōu)異的活性(1 M KOH/堿化海水中分別為 230/258 mV@100 mA cm-2)及1500小時(shí)以上的穩(wěn)定性。組裝的AEMWE(CoPO||R-PO4-NiCoFeOOH)在較低的1.81 V電池電壓即可提供1.0 A cm-2電流密度,同時(shí)在500 mA cm-2下可持續(xù)運(yùn)行300小時(shí),并可由可再生能源驅(qū)動(dòng),顯示出其良好的工業(yè)應(yīng)用潛力。
圖文詳解:
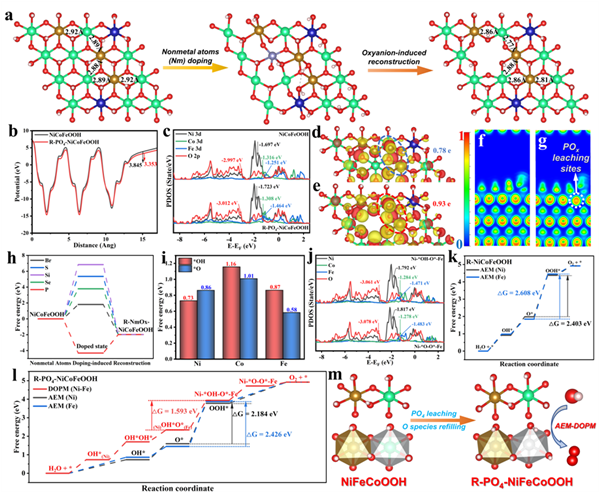
Figure 1. Thoretical ideas. (a) Theoretical structural models of (001) crystal planes in NiCoFeOOH, PO4-NiCoFeOOH, and R-PO4-NiCoFeOOH and corresponding Ni-Fe atom distances. (b) The calculated WF curves of NiCoFeOOH and R-PO4-NiCoFeOOH models. (c) Calculated PDOS of Ni, Fe, Co, and O atoms of NiCoFeOOH and R-PO4-NiCoFeOOH models. The DCD of (d) NiCoFeOOH and (e) R-PO4-NiCoFeOOH (Red color is related to the electron acceptance; yellow color is related to the electron donation). The ELF of (f) NiCoFeOOH and (g) R-PO4-NiCoFeOOH models. (h) The formation energy of various ions doping NiCoFeOOH models. (i) The formation energy of *O and *OH at Ni, Fe, and Co sites for R-PO4-NiCoFeOOH. (j) The PDOS of Ni, Fe, Co, and O atoms in Ni-*OH-O*-Fe and Ni- *O-O*-Fe intermediates for R-PO4-NiCoFeOOH. (k) Gibbs free energy diagrams of the AEM pathway at Ni and Fe sites for R-NiCoFeOOH. (l) The Gibbs free energy diagrams of the AEM pathway at Ni and Fe sites, as well as the DOPM pathway at Ni-Fe dual-sites for R-PO4-NiCoFeOOH. (m) Schematic diagram of PO4-induced deep reconstruction for realizing the DOPM pathway.
首先,通過密度泛函理論計(jì)算分析了氧陰離子誘導(dǎo)羥基氧化物的結(jié)構(gòu)改善與催化行為。PO43-浸出誘導(dǎo)了Ni/Fe-O6八面體塌縮,其Ni-O-Fe金屬間距更近,有利于實(shí)現(xiàn)中間體耦合(圖1a)。同時(shí),相應(yīng)WF、DOS、DCD、ELF等計(jì)算表明了PO43-誘導(dǎo)了電子離域化的高價(jià)Ni-Fe雙位點(diǎn)形成(圖1b-h)。結(jié)合中間體吸附模型計(jì)算證明了Ni與Fe位點(diǎn)分別對(duì)于*OH與*O的吸附更有利,有望實(shí)現(xiàn)差異性Ni-OH-O-Fe中間體耦合(圖1i-k)。此外,基于AEM與DOPM路徑計(jì)算了NiCoFeOOH與R-PO4-NiCoFeOOH模型的吉布斯吸附自由能臺(tái)階圖。R-PO4-NiCoFeOOH在Ni與Fe位點(diǎn)上的決速步驟能壘更低,同時(shí)觸發(fā)了更有利的DOPM路徑,極大改善了催化活性與穩(wěn)定性。
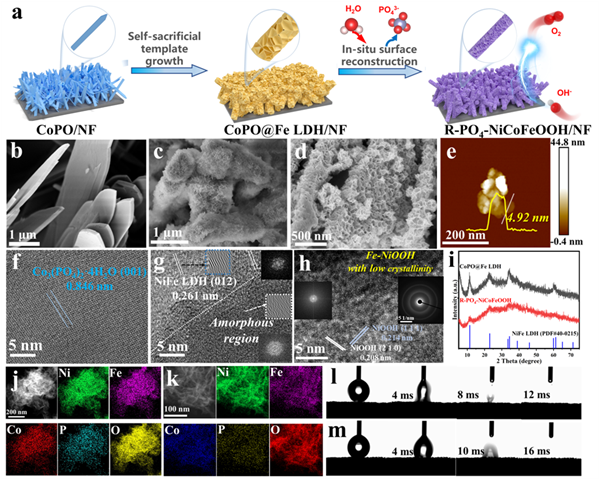
Figure 2. Preparation and structures of catalysts. (a) Synthetic route of heterostructure R-PO4-NiCoFeOOH, with the inset zoomed-in images presenting the surface structure evolution of the microflakes/nanosheets. The SEM images of (b) CoPO, (c) CoPO@Fe LDH, and (d) R-PO4-NiCoFeOOH. (e) AFM images of R-PO4-NiCoFeOOH. The HRTEM images of (f) CoPO, (g) CoPO@Fe LDH, and (h) R-PO4-NiCoFeOOH. Inset, the SAED images. (i) The XRD patterns of CoPO@Fe LDH precursors and R-PO4-NiCoFeOOH actives. (j,k) The corresponding EDS element mappings of R-PO4-NiCoFeOOH. The water contact angle measurements of (l) R-PO4-NiCoFeOOHand (m) R-NiCoFeOOH.
隨后,進(jìn)行了催化劑的合成、結(jié)構(gòu)表征與電化學(xué)重構(gòu)行為研究。CoPO@Fe LDH/NF前驅(qū)體有著粗糙的異質(zhì)表面,表現(xiàn)出LDH納米片-磷酸鈷微米片-三維NF基底的分級(jí)結(jié)構(gòu)(圖2a-c)。深度重構(gòu)后P物種信號(hào)減弱,XRD與TEM表現(xiàn)出無定形羥基氧化物物相。結(jié)合相應(yīng)的XPS、Raman與電化學(xué)分析等證明了其原位深度重構(gòu)過程(圖2d-i)。此外,R-PO4-NiCoFeOOH表現(xiàn)出均勻的元素分布與親水性(圖2j-m)。
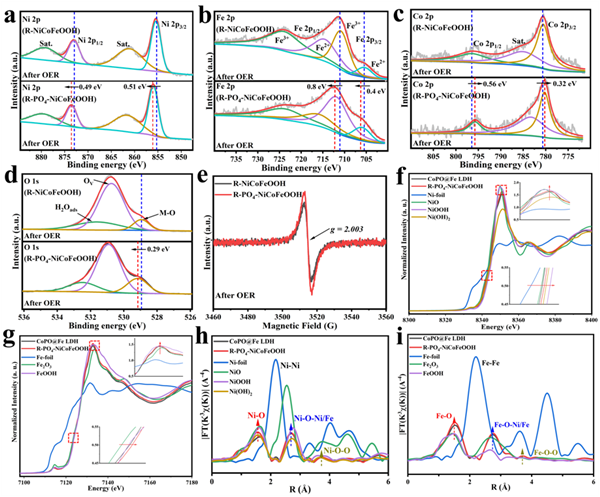
Figure 3. Electronic and coordination structures of the catalysts. The XPS spectra of (a) Ni 2p, (b) Fe 2p, (c) Co 2p, and (d) O 1s for R-NiCoFeOOH and R-PO4-NiCoFeOOH. (e) EPR spectra of samples. The XANES of (f) Ni and (g) Fe K-edges spectra for all the samples. The FT-EXAFS spectra of (h) Ni and (i) Fe K-edges for the samples.
通過XPS與XAS研究了催化劑電化學(xué)活化重構(gòu)前的電子與配位結(jié)構(gòu)變化。R-PO4-NiCoFeOOH的Ni和Fe 2p光譜在CV重構(gòu)后均向高結(jié)合能方向偏移,表明其OER活化過程后的價(jià)態(tài)升高(圖3a-c)。O的XPS與EPR也證明了其更完全的重構(gòu)行為(圖3d-e)。XAS表明活化后的具有更近的Ni(Fe)-O-Ni(Fe)的距離與更飽和的Ni(Fe)-O配位環(huán)境,并有著更強(qiáng)的M-O-O路徑峰,有利于實(shí)現(xiàn)O-OH氧中間體直接耦合(圖3g-i)。
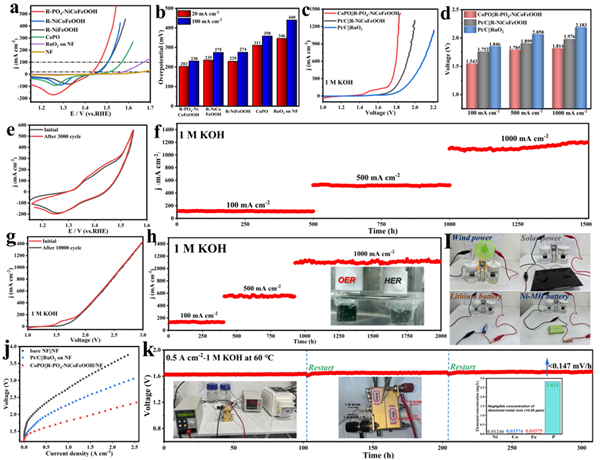
Figure 4. Electrocatalytic performance of catalysts. (a) OER polarization curves of R-PO4-NiCoFeOOH and other samples in 1 M KOH solutions and (b) the overpotential diagram (@20 and 100 mA cm-2) corresponding to the polarization curves of OER. (c) Polarization curves of Pt/C||R-NiCoFeOOH, Pt/C||RuO2, and CoPO||R-PO4-NiCoFeOOH for overall water splitting with 90% iR compensation. (d) The cell voltage diagram (@100, 500, and 1000 mA cm-2) corresponding to the polarization curves of overall water splitting. (e) The CV curves of R-PO4-NiCoFeOOH/NF before and after 3000 CV cycles in 1 M KOH solutions. (f) Time-reliant current density curves of R-PO4-NiCoFeOOH under several invariant overpotentials for over 1500 h in 1 M KOH solutions. (g) LSV curves of CoPO||R-PO4-NiCoFeOOH before and after 10000 CV cycles in 1 M KOH solutions. (h) Long-term tests of water electrolysis with the CoPO||R-PO4-NiCoFeOOH system. Inset: the digital photograph of the anode and cathode during the overall water-splitting process. (i) Actual operation diagram of water splitting powered by wind, sunlight nickel metal hydride (NMH), and lithium cell. (j) Polarization curves of the AEMWE obtained at 60 °C with Fumasep FAA-3-PK-130 membrane. (k) Chronopotentiometry curves of the CoPO||R-PO4-NiCoFeOOH based AEM electrolyzer. Inset: practical devices of AEM electrolyzer and ICP test of Ni, Co, Fe, and P elements in the electrolytes after stability test.
進(jìn)一步測(cè)試了催化劑的電化學(xué)性能。R-PO4-NiCoFeOOH在1 M KOH與堿化海水中分別只需要230與258 mV的超低過電位即可達(dá)到100 mA cm-2的電流密度,且具有3000圈CV循環(huán)與1500小時(shí)以上的長期穩(wěn)定性,并可由可再生能源驅(qū)動(dòng)(圖4a-i)。此外,其在全解水系統(tǒng)與AEMWE中也有著優(yōu)異性能,60 ℃下CoPO||R-PO4-NiCoFeOOH在1 A cm-2電流密度下僅需較低的槽壓(1.81 V),并實(shí)現(xiàn)了超過300小時(shí)的穩(wěn)定運(yùn)行(圖4j,k)。電化學(xué)測(cè)試結(jié)果顯示了其良好的商業(yè)應(yīng)用潛力。

Figure 5. OER catalytic mechanism. In-situ Raman spectra for (a) CoFe LDH/NF and (b) CoPO@Fe LDH/NF in 1 M KOH solutions. (c) Corresponding peak intensity and position change with the applied potential in the in situ Raman spectra. The in situ ATR-FTIR spectra of (d) R-NiCoFeOOH, (e) R-PO4-NiCoFeOOH, and (f) 18O-labeled R-PO4-NiCoFeOOH. Detected DEMS signals without any correction or subtraction of 16O16O, 16O18O, and 18O18O for (g) R-PO4-NiCoFeOOH and (h) R-NiCoFeOOH relative to time after activation in 18O-labeled KOH solutions. (i) The 36O2/34O2 evolution mechansim of R-PO4?NiCoFeOOH. Schematic illustration of (j) PO43--optimized AEM pathway and (k) PO4 leaching-triggered DOPM pathway.
最后,通過原位光譜/質(zhì)譜探究了其內(nèi)在催化機(jī)制及本征活性起源。原位拉曼光譜(圖5a-c)表明了R-PO4-NiCoFeOOH中無序態(tài)更強(qiáng)的γ-NiOOH物相的存在;此外,NiOOH特征峰的延遲出現(xiàn)暗示了其緩慢深度的重構(gòu)過程。原位紅外光譜(圖5d-f)中,R-PO4-NiCoFeOOH表現(xiàn)出額外的*O、*OH以及*O-OH*的振動(dòng)峰,表明其一定的差異性吸附物種耦合路徑的存在。從電化學(xué)質(zhì)譜可以看出(圖5g-i),18O標(biāo)記的R-PO4-NiCoFeOOH表現(xiàn)出更強(qiáng)的34O2和36O2信號(hào),暗示更強(qiáng)烈的離子浸出與O填充過程,實(shí)現(xiàn)了催化劑表面吸附18O物種的標(biāo)記。同時(shí),表面的吸附18OH基團(tuán)參與了直接的氧氧耦合而脫出形成36O2,表明R-PO4-NiCoFeOOH除了遵循典型的AEM機(jī)制以外,還有額外的雙位點(diǎn)DOPM機(jī)制的參與(圖5j,k)。
總結(jié):
本文在理論指導(dǎo)下,設(shè)計(jì)了以磷酸鈷模板合成的NiFe LDH預(yù)催化劑,通過磷酸根離子摻入促進(jìn)其深度重構(gòu)為R-PO4-NiCoFeOOH活性催化劑,調(diào)節(jié)了其原子排列與電子分布,有利于觸發(fā)協(xié)同雙位點(diǎn)協(xié)同機(jī)制。即利用Ni/Fe位點(diǎn)不同的*OH/*O傾向性,實(shí)現(xiàn)路徑最優(yōu)的Ni-*OH-O*-Fe自由基耦合,從而加快催化反應(yīng)動(dòng)力學(xué)。實(shí)驗(yàn)證明了其優(yōu)化的雙位點(diǎn)差異性氧化路徑(DOPM)機(jī)制的形成,以及對(duì)提高催化劑OER活性與穩(wěn)定性的重要作用,并在AEMWE中表現(xiàn)出良好的工業(yè)應(yīng)用潛力(60 ℃下1.81 [email protected] A cm-2,0.5 A cm-2@300小時(shí))。該研究為合理設(shè)計(jì)高效穩(wěn)定的非貴金屬基催化劑的雙位點(diǎn)催化機(jī)制提供了新思路。
文獻(xiàn)信息:
Jiang, L.; Chen, X.; Jiang, L.; Luo, X.; Li, R.; Zhou, Q.; Mu, X.; Chen, L.; Yu, J.; Mu, S. Oxyanion-Triggered Discrepant Oxide Pathways Via Tailoring Reconstruction of Non-Noble Catalysts for Water Oxidation. Nano Lett. 2025.
https://pubs.acs.org/doi/10.1021/acs.nanolett.5c03709
作者簡(jiǎn)介:
木士春教授簡(jiǎn)介:武漢理工大學(xué)首席教授,博士生導(dǎo)師,國家級(jí)高層次人才。長期致力于質(zhì)子交換膜燃電池和電解水催化劑及鋰電池關(guān)鍵材料研發(fā)。以第一作者或通訊作者在Nat. Commun.、Adv. Mater.、J. Am. Chem. Soc.、Angew. Chem. Int. Ed.、Energy Environ. Sci.、Nano Lett.等國內(nèi)外期刊上發(fā)表320余篇高質(zhì)量學(xué)術(shù)論文。H 因子104(WOS);申請(qǐng)國家發(fā)明專利112余件,其中授權(quán)86件;獲國家技術(shù)發(fā)明獎(jiǎng)二等獎(jiǎng)1項(xiàng)。
課題組介紹:
武漢理工大學(xué)先進(jìn)能源材料研究團(tuán)隊(duì)依托材料復(fù)合新技術(shù)國家重點(diǎn)實(shí)驗(yàn)室,長期從事質(zhì)子交換膜燃料電池關(guān)鍵材料與核心器件、電化學(xué)產(chǎn)氫催化材料、鋰離子電池電極材料和碳納米材料等研究工作。歡迎有志于科技報(bào)國的研究生及博士后加入團(tuán)隊(duì)!
木士春研究團(tuán)隊(duì)主頁:http://shichunmu.polymer.cn/