- Prof. Dr. Sheng Zhang
- Beijing University of Chemical Technology
- Welcome to zhangshengfrml.polymer.cn
- Visits:1051154
Links
Contact Info.
- Address:北京市朝陽區北三環東路15號2號信箱
- Zip:100929
- Tel:010-64436820
- Fax:010-64436820
- Email:[email protected]
Current Location :> Home > Publications > Text
Effects of Surface Acid-Activated Kaolinite on the Fire Performance of Polypropylene Composite.
writer:唐武飛,張勝,孫軍, 李洪飛, 劉曉東, 谷曉昱
keywords:the Fire Performance of Polypropylene Composite
source:期刊
Issue time:2016年
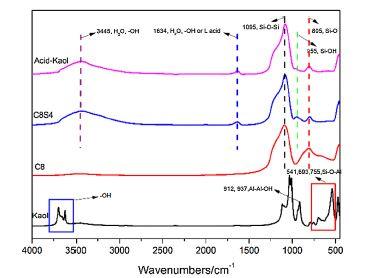 Modified kaolinite containing Lewis acidic sites (Acid-Kaol) was prepared by calcination of kaolinite (Kaol) and successive acid treatment. Acid-Kaol was then introduced into polypropylene (PP) with intumescent flame-retardant (IFR). The flammability and thermostability of PP/IFR/Acid-Kaol were studied by limiting oxygen index (LOI), vertical buring test (UL-94), cone calorimeter test (CCT) and thermogravimetric analysis (TGA). LOI value ofPP/25 wt.% IFR composite was 31.1%, it increased to 34.9% by 1.5 wt.% Acid-Kaol substitution with IFR. Moreover,the peak of heat release rate (PHRR) of PP/IFR/Acid-Kaol decreased 46.8% compared with that of PP/IFR composite.The residues after CCT were characterized by scanning electron microscopy (SEM), X-ray photoelectron spectrometer (XPS) and FTIR. It was indicated that Acid-Kaol accelerated crosslinking during combustion and promoted char formation, which will act as insulating barrier to protect the matrix from further decomposed. © 2016 Elsevier B.V.
Modified kaolinite containing Lewis acidic sites (Acid-Kaol) was prepared by calcination of kaolinite (Kaol) and successive acid treatment. Acid-Kaol was then introduced into polypropylene (PP) with intumescent flame-retardant (IFR). The flammability and thermostability of PP/IFR/Acid-Kaol were studied by limiting oxygen index (LOI), vertical buring test (UL-94), cone calorimeter test (CCT) and thermogravimetric analysis (TGA). LOI value ofPP/25 wt.% IFR composite was 31.1%, it increased to 34.9% by 1.5 wt.% Acid-Kaol substitution with IFR. Moreover,the peak of heat release rate (PHRR) of PP/IFR/Acid-Kaol decreased 46.8% compared with that of PP/IFR composite.The residues after CCT were characterized by scanning electron microscopy (SEM), X-ray photoelectron spectrometer (XPS) and FTIR. It was indicated that Acid-Kaol accelerated crosslinking during combustion and promoted char formation, which will act as insulating barrier to protect the matrix from further decomposed. © 2016 Elsevier B.V.
keywords:the Fire Performance of Polypropylene Composite
source:期刊
Issue time:2016年
 Modified kaolinite containing Lewis acidic sites (Acid-Kaol) was prepared by calcination of kaolinite (Kaol) and successive acid treatment. Acid-Kaol was then introduced into polypropylene (PP) with intumescent flame-retardant (IFR). The flammability and thermostability of PP/IFR/Acid-Kaol were studied by limiting oxygen index (LOI), vertical buring test (UL-94), cone calorimeter test (CCT) and thermogravimetric analysis (TGA). LOI value ofPP/25 wt.% IFR composite was 31.1%, it increased to 34.9% by 1.5 wt.% Acid-Kaol substitution with IFR. Moreover,the peak of heat release rate (PHRR) of PP/IFR/Acid-Kaol decreased 46.8% compared with that of PP/IFR composite.The residues after CCT were characterized by scanning electron microscopy (SEM), X-ray photoelectron spectrometer (XPS) and FTIR. It was indicated that Acid-Kaol accelerated crosslinking during combustion and promoted char formation, which will act as insulating barrier to protect the matrix from further decomposed. © 2016 Elsevier B.V.
Modified kaolinite containing Lewis acidic sites (Acid-Kaol) was prepared by calcination of kaolinite (Kaol) and successive acid treatment. Acid-Kaol was then introduced into polypropylene (PP) with intumescent flame-retardant (IFR). The flammability and thermostability of PP/IFR/Acid-Kaol were studied by limiting oxygen index (LOI), vertical buring test (UL-94), cone calorimeter test (CCT) and thermogravimetric analysis (TGA). LOI value ofPP/25 wt.% IFR composite was 31.1%, it increased to 34.9% by 1.5 wt.% Acid-Kaol substitution with IFR. Moreover,the peak of heat release rate (PHRR) of PP/IFR/Acid-Kaol decreased 46.8% compared with that of PP/IFR composite.The residues after CCT were characterized by scanning electron microscopy (SEM), X-ray photoelectron spectrometer (XPS) and FTIR. It was indicated that Acid-Kaol accelerated crosslinking during combustion and promoted char formation, which will act as insulating barrier to protect the matrix from further decomposed. © 2016 Elsevier B.V.