�S�����ܼ����Ϳɴ�������O��Ŀ��ٰlչ����Ό��F�������ְ�ȫ�ĸ�������Դ�惦�ɞ����ǰ�ص���Ҫ�n�}�����գ��㽭������W�����ƌW�c���̌WԺ�����H�z�I�WԺ��������ڈF��ڇ��H�����ڿ���Nano Energy����Ӱ�����16.8���ھ��l�����}�顶Janus-structured composite nanofiber membranes with heterointerfacial engineering for high-performance all-solid-state lithium metal batteries�����о�Փ�ġ�ԓ�о������һ�N�������S��������Y����Janus�ͺϼ{���w�SĤ���@�������˹̑B䇽���늳ص��x�ӂ�ݔЧ�ʺͽ��淀���ԣ����������ܼ��������еă��܆��}�ṩ�˄����ԵĽ�Q������Փ��DOI��10.1016/j.nanoen.2025.111136�����ĵ�һ���ߞ��㽭������W�����ƌW�c���̌WԺ�����H�z�I�WԺ����ʿ�о�������A��ͨӍ���ߞ��㽭������W��ʿ����������������

����������O��߰�ȫ����ϵ�yѸ�ٰlչ�ı����£��̑B�늳����䃞���ğ᷀�����c������ȫ�ԣ�����u�ɞ���һ��늳ؼ��g�ĺ��ķ���Ȼ������һ�o�C�̑B늽��|�mȻ����ԏ����s�������y���c�������������ܛ�|������ƥ�䣻�����ۺ���늽��|�mȻ��ܛ�ӹ������x�ӌ���Բ��㣬�y�ԝM����������ͺϹ̑B늽��|�ںϟo�C�{���w���ĸߌ�����c�ۺ�������g�ԣ��ɞ錍�F��Ч�Ұ�ȫ���ܵ��������������y�о�����ö��S�Ӡ�Y�������䮐�|������e���ޣ��Cе���Ч�����㣬�Ƽs�˲��������ܼ����������I��ďV�����á�
��ˣ�����Fꠄ��²���늼����g�������˾������S����Janus���|����ďͺϼ{���w�SĤ��ԓ�Y������ܛ��PVDF-HFP�����϶���̶�LLZO�{���w�������Y���ܻ��������ۺ���Y���ȣ����F���x�ӂ�ݔ·���Ķ�Ԫ���ͽ��������ݵă�����ԓ�OӋ���H�@��������Ĥ�ęCе���ȣ��_��4.24 MPa����߀���F��7.60×10-4 S·cm-1��50��C�����x�ӌ���ʺ�0.75����x���w�Ɣ�����ՓӋ���@ʾ��JanusĤ�����C-F���܈F��·��˹��A����λ�c��Ч���M��}���x��������x���w���܉����γɷ�����Li3N/LiF����SEI�ӣ����C��䇽��ٵľ�����e���L����ѭ�h��ԓ�ͺ�늽��|�ڌ��H����ܛ��늳���չ�F���������ܣ��܉��ӿ����ϵ����l���b����ACEL���ͼ�늂���������180���ۯB��ᘴ̡��Cе���м��ߜحh������Ȼ���������������������ܼ��������ԃ��ܵ�����ԓ�о����H�IJ��ϽY���OӋ����ͻ���˂��y�̑B늽��|�ڙCе���Ⱥ��x�ӂ�ݔ֮�g��ì�ܣ��������ܼ����I�����Դ�����ṩ�ˏ������ļ��g֧�Ρ�
���w���ԣ�ԓ�о�������һ�NJanus�Y���ďͺ��w�SĤ��ͨ�^��PEO���w�Ȳ��������|���棬�ṩ�˶����x�ӂ�ݔ·�����@�NJanus�Y���ďͺ�늽��|��PHSL-CSE����ͨ�^�o늼��z���g�������S�����W�j���F�ġ��@�NJanus�Y����PEO-LLZO���昋�����S���ğo�C-�ۺ��ﮐ�|���棬�{�����x�ӂ�ݔ�О飬�γ��˶�N��x�ӂ�ݔͨ����(i) PEO���w�ȵ�朶��\�ӣ�(ii) LLZO�w���γɵ��B�m�ࣻ(iii) PEO-LLZO���|����Ŀ��g늺Ɍӡ��@Щ�C�ƅfͬ���ã��@���������x����늽��|�ȵĂ�ݔЧ�ʡ�

Fig. 1. Schematic illustration of the ion transport mechanism and fabrication process of the PHSL-CSE composite solid electrolyte with a three-dimensional interconnected Janus structure.(a) Representation of the Janus-structured composite fiber membrane, showcasing its unique heterogeneous interface and architecture.(b) Illustration of the multiple lithium-ion transport pathways enabled by the Janus structure, highlighting the role of the interconnected network in facilitating efficient ion migration. (c)Schematic of the full-cell configuration assembled with the PHSL-CSE composite solid-state electrolyte, demonstrating its integration into a practical solid-state lithium battery. (d) Step-by-step fabrication process of the PHSL-CSE composite electrolyte, emphasizing the formation of the Janus structure and the incorporation of functionalized components.
ͨ�^X�侀���䣨XRD����������@�R��TEM������������@�R��SEM����Ԫ��ӳ����ֶΌ������M���˱������Y���C����Janus�Y���ijɹ������Լ�LLZO�w�����w�S����̓Ȳ��ľ���ֲ����@�_�����B�m���x��ͨ�������õĽ��������ԡ������~׃�Q�t����V��FTIR������������LLZO�ļ��뽵����PVDF-HFP�ĽY���ȣ�LLZO��SN�fͬ���M��PVDF-HFP��Ó�����䷴������ʾ�������ᷨ��DSC���@ʾ��Janus�Y���{���w�S�������@��������PEO朶εĽY�������ط�����TGA���ߜyԇ������PHSL-CSE늽��|���Ѓ����ğ᷀���Ժͳߴ緀���ԡ��Cе���ܜyԇ�@ʾ��ԓ늽��|���иߙCе���ȣ�4.24 MPa�������õ����g�ԡ�
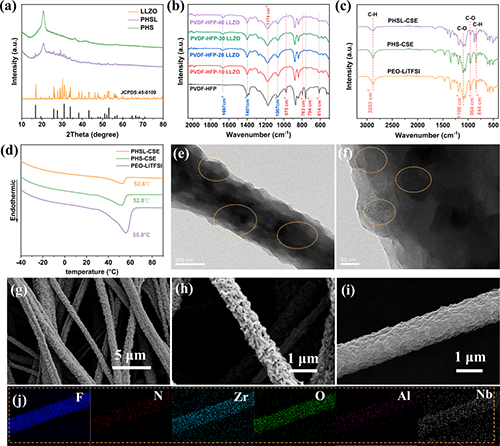
Fig. 2. Comprehensive characterization of the Janus fiber membrane and electrolytes across different systems. (a) X-ray diffraction (XRD) patterns of LLZO, PHS-CSE, and PHSL (Janus) composite fiber membranes. (b) Attenuated total reflection Fourier-transform infrared (ATR-FTIR) spectra of PHSL fibers with varying LLZO content. (c) ATR-FTIR spectra of PEO-LiTFSI, PHS-CSE, and PHSL-CSE solid electrolyte films. (d) Differential scanning calorimetry (DSC) curves of PEO-LiTFSI, PHS-CSE, and PHSL-CSE solid electrolytes. (e, f) Transmission electron microscopy (TEM) images of PHSL (Janus) composite nanofibers at different magnifications. (g, h) SEM images of PHSL (Janus) composite nanofibers at varying magnifications. (i) SEM image of a single Janus fiber (j) Corresponding elemental mapping of the Janus fiber, illustrating the spatial distribution of key elements and confirming the structural heterogeneity of the Janus architecture.
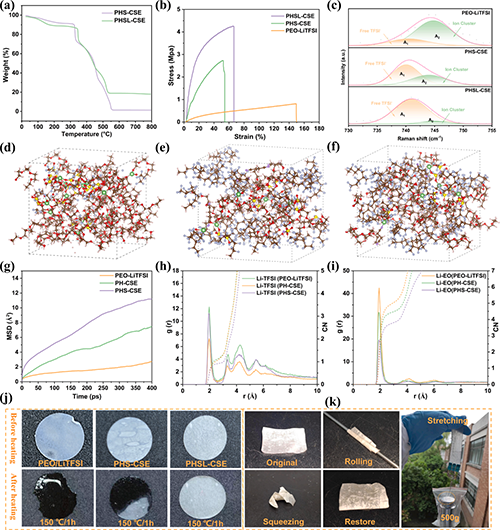
Fig. 3. Mechanical and thermal properties of composite solid electrolytes and their molecular coordination mechanisms. (a) TGA curves of PHS-CSE and PHSL-CSE composite solid electrolytes. (b) Stress-strain curves of PEO-LiTFSI, PHS-CSE, and PHSL-CSE composite solid electrolytes. (c) Raman spectra of PEO-LiTFSI, PHS-CSE, and PHSL-CSE composite solid electrolytes within the 730�C755cm-1 range. (d�Cf) Schematic molecular coordination models of (d) PEO-LiTFSI, (e) PH-CSE, and (f) PHS-CSE electrolytes. (g) Mean square displacement (MSD) profiles of lithium ions in PEO-LiTFSI, PH-CSE, and PHS-CSE electrolytes. (h) Radial distribution functions (RDF) and coordination numbers (CN) for Li+and TFSI-1 in PEO-LiTFSI, PH-CSE, and PHS-CSE electrolytes. (i) RDF and CN for Li+ and thylene oxide (EO) segments in PEO-LiTFSI, PH-CSE, and PHS-CSE electrolytes. (j) Digital photographs of PEO-LiTFSI, PHS-CSE, and PHSL-CSE composite solid electrolytes before and after heating at 150��C for 30minutes. (k) Digital images showcasing the mechanical robustness of PHSL-CSE solid electrolyte films in various states, including original, curled, compressed, recovered, and in a suspended state under a 500g weight.
늻��W���ܷ��棬PHSL-CSE늽��|��50��C�r�x��늌����_��7.60 × 10-4 S cm-1����x���w�Ɣ���0.75��Arrhenius�D�@ʾ�����^�͡��������V���ܶȷ�����Փ��DFT��Ӌ�������PVDF-HFP��TFSI-��x�����^���������ã����M��}���x��������x���w�ơ����ӄ����W��MD��ģ�MҲ֧������x���ڏͺ�늽��|�и���ĔUɢ�����Ԓ����������LSV���@ʾ늻��W���ڌ��_4.94 V��

Fig. 4.Electrochemical performance of composite solid electrolytes. (a) Electrochemical impedance spectroscopy (EIS) spectra of the PHSL-CSE composite solid electrolyte at various temperatures. (b) Arrhenius plots of ionic conductivity for PEO-LiTFSI, PHS-CSE, and PHSL-CSE composite solid electrolytes. (c) Chronoamperometric response of Li | PHSL-CSE | Li symmetric cell; inset: Nyquist impedance spectra recorded before and after polarization. (d) Linear sweep voltammetry (LSV) curves of PEO-LiTFSI, PHS-CSE, and PHSL-CSE composite solid electrolytes. (e) Energy level diagrams illustrating the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) for PVDF-HFP, PEO, LiTFSI, and succinonitrile (SN). (f) Binding energies of TFSI- with SN, PEO, and PVDF-HFP. (g) Critical current density (CCD) curves of PEO-LiTFSI, PHS-CSE, and PHSL-CSE composite solid electrolytes. (h) Interfacial resistance of the Li | PHSL-CSE | Li symmetric cell before and after 100 charge-discharge cycles. (i) Tafel plots of PEO-LiTFSI, PHS-CSE, and PHSL-CSE composite solid electrolytes.
�c䇽���ؓ�O�Ľ��淀���ԃ�����Li/Li���Q늳�ѭ�h�����Գ��^3500С�r���@������Janus�Y�����M�γ��˸���Li3N/LiF�ķ���SEI�ӡ�XPS������DFTӋ���C����SEI�ijɷּ��䌦����䇳��e������֦�����L�����á�XPS����������ѭ�h���Li | PHSL-CSE | Li늳����ؓ�O�����γ��˸���Li3N/LiF��SEI�ӡ�DFTӋ�㣨�����ܣ��C����Li+����������LiF��Li3N���棬�������֦�����S�C�κ˺����L���@�N������SEI����Ч���x��늽��|�c䇽��ٵ�ֱ�ӽ��|���p���˸�������
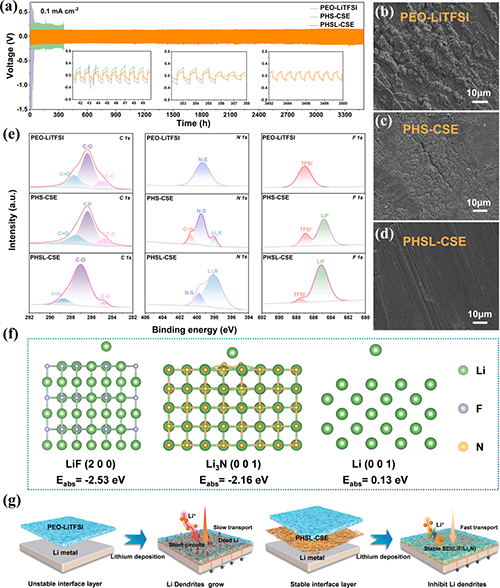
Fig. 5. Long-term cycling performance and morphological characterization of lithium metal surfaces.(a) Cycling stability of Li/Li symmetric cells with PEO-LiTFSI, PHS-CSE, and PHSL-CSE composite solid electrolytes at a current density of 0.1mAcm-2. (b-d) SEM images of lithium metal surfaces after 50 cycles for Li | PEO-LiTFSI | Li cells, and 100 cycles for Li | PHS-CSE | Li and Li | PHSL-CSE | Li cells. (e) XPS spectra of the lithium metal surfaces after cycling.(f) Adsorption energies of Li+ on LiF (200), Li3N (001), and Li (001) interfaces. (g) Schematic representation of the lithium deposition mechanism using PEO-LiTFSI and PHSL-CSE composite solid electrolytes.
ԓ늽��|��ȫ늳���Ҳ���F��ɫ��LiFePO4 | PHSL-CSE | Li늳���0.5 C��ѭ�h500�κ������������Ժܸߡ�NCM811 | PHSL-CSE | Li늳���1 C��ѭ�h100�κ�Ҳ���������õ�����������Ҫ���ǣ�����PHSL-CSE늽��|������ܛ��늳�չ�F��Խ�ļ��������͙Cе�����ԡ���ʹ��180���ۯB���Cе���С�ᘴ̺�80��C�ᱩ¶�ȘO�˗l���£�ܛ��늳����ܷ�����ACEL�l�������ͼ�늂�������늣��@ʾ�����ڿɴ�������O���I��ľ�����

Fig. 6. Performance evaluation of all-solid-state lithium metal batteries at 50��C. (a) Electrochemical impedance spectroscopy (EIS) of Li | PEO-LiTFSI | LiFePO4, Li | PHS-CSE | LiFePO4, and Li | PHSL-CSE | LiFePO4 cells. (b) Voltage versus areal capacity curves for Li | PHSL-CSE | LiFePO4 cells. (c) Long-term cycling performance of Li | PHSL-CSE | LiFePO4 cells at a 0.2C rate. (d) Rate performance of Li | PHSL-CSE | LiFePO4 cells. (g) Voltage versus areal capacity curves at various rates for Li | PHSL-CSE | LiFePO4 cells. (e) Voltage versus areal capacity curves and (f) long-term cycling performance of Li | PHSL-CSE | LiFePO4 cells at a 0.5?C rate. (h) Voltage versus areal capacity curves and (i) long-term cycling performance of Li | PHSL-CSE | NCM811 cells at a 1C rate. (j) Optical images of Li | PHSL-CSE | LiFePO4 solid-state pouch lithium metal batteries, showing stable power output under various mechanical deformations including flat, bent, folded, cut, and punctured states.(k) A solid-state pouch cell integrated with an EMG armband, demonstrating the pouch battery''s capability to power a small vehicle via a flexible interface. (l) The pouch battery maintains stable output voltage even in a folded state. (m) Integration of the pouch battery with an ACEL flexible display, providing stable power to a flexible electronic device.
������֮���@��о�ͨ�^Janus�Y���ͺϼ{���w�SĤ�Į��|���湤�̣��ɹ��_�l��һ�N�����ܡ����Ե�ȫ�̑B늽��|��ԓ늽��|�����x�ӌ���ʡ����w�Ɣ��������ęCе/�᷀���Ժͽ��淀������һ�w����Ч�������֦�����L����������늳ؑ�����չ�F���������@헹������_�l���ڃ��ܺͿɴ�������O��ĸ����ܡ�����ȫ�̑B�늳��ṩ����˼·��
�ڴˣ����x�㽭ʡ��Ȼ�ƌW�����Ŀ��LY21E030023�����㽭������W���݄����о�Ժ�����Ŀ��SYY2024C000008����֧�֣�
ͨӍ���ߺ��������㣬�У���ʿ�����ڣ���ʿ���������㽭������W�����ƌW�c���̌WԺ�����H�z�I�WԺ����Ժ�L����Ҫ���·�ˮ���|Ⱦ���¼��g������������ܼ���Ʒ�о����Ե�һ����ͨӍ������ Advanced Functional Materials, Nano Letters, Energy Storage Materials, Nano Energy, Chemical Engineering Journal�ȿ����ϰl��SCIՓ��70��ƪ���ڙ���D�����Ұl������30��헡��@�Ç��Ҽ��̌W�ɹ����Ȫ����㽭ʡ�̌W�ɹ��صȪ���1헣����֫@���Ї��������I�ϕ��̌W�ɹ�һ���������Ȫ����㽭ʡ��Ȼ�ƌW�����Ȫ����Ї��̘I�ϕ��Ƽ��M�������Ȫ���1헡�
ԭ��朽ӣ�https://doi.org/10.1016/j.nanoen.2025.111136
- �����Janus�w�����p�ɳɞ��{�Ǝ� - ����/���A�����ΈF��B�lNature�ӿ����༉Janus�Y������������ 2026-02-06
- ���ƴ�Ȕ���ڡ���cϲ���ڈF� CEJ����Ч��ܵ�Janus����{�ͺ�Ĥ���ڿɿصĸ��`��ˮ���Ƅ��� 2023-10-27
- ���_��īԴ/���������ɡ�Adv. Sci.�������Janus�Y��������ܵ����ܳ������ 2023-05-31
- ��ɽ��W��־�x�����w�����/UCLA�R�������� Sci. Adv.���ɿ��١�Ҏģ���Ƃ�ĸߏ��g�����x�����zĤ���ڸ���������������� 2026-03-03
- �㹤��������/�~��Ҋ�n�}�M Macromolecules��ͨ�^��ΑB�OӋ��Ч�Ƃ�����ܿɻ����zճ�� 2026-03-01
- ��ƴ���������־�� Adv. Mater.�����ÿs������ӑB�I�����ӑB���r-�����Ӆfͬ�W�j ���F�����ܿɻ��՟���Ԙ�֬ 2026-02-13
- ������������� Adv. Funct. Mater.�������Y���ͺ�늽��|��ȫ�̑B䇽���늳��ṩ��·�� 2025-09-03
