�S�����ܼ����Ϳɴ�������O(sh��)���Ѹ�Ͱl(f��)չ����Ό������ܵ���Դ�惦��Ԫ���Ի������ɻ����ѳɞ鼏�������c��Դ�ƌW(xu��)�����I(l��ng)���ǰ���n�}�����y(t��ng)��Ӳ�|(zh��)Һ�B(t��i)늳��y�ԝM�㿗���ڏ������ۯB���������B(t��i)�µ����������գ��㽭������W(xu��)�����ƌW(xu��)�c���̌W(xu��)Ժ�����H�z�I�W(xu��)Ժ��������ڈF�������Ȼ���ӵĽY(ji��)��(g��u)�м�ȡ�`�У��ɹ��_�l(f��)��һ�N������(ji��)�Y(ji��)��(g��u)��(f��)�Ϲ̑B(t��i)늽��|(zh��)���@헄�(chu��ng)�����о����w�S�Y(ji��)��(g��u)�c늳�늽��|(zh��)����Y(ji��)�ϣ������Կɴ��������ṩ��ȫ�µĽ�Q������ԓ�ɹ��ԡ�Biomimetic Bamboo-Node-Inspired Composite Electrolyte with Hierarchical Ion Pathways for Safe and High-Performance Solid-State Lithium Metal Batteries�����}���ڇ��H��(qu��n)���ڿ���Advanced Functional Materials����Ӱ�����19.0���ϰl(f��)����Փ�ĵ�һ���ߞ��㽭������W(xu��)�����ƌW(xu��)�c���̌W(xu��)Ժ�����H�z�I�W(xu��)Ժ����ʿ�о�������A��ͨӍ���ߞ��㽭������W(xu��)��ʿ����(d��o)���������ڡ�

�S����yʽ����O(sh��)������ܼ���Ʒ�Ŀ��ٰl(f��)չ������߸߰�ȫ�ԡ��������ܶȺ��L����������늳������������С�ȫ�̑B(t��i)䇽���늳أ�SSLMBs��������еİ�ȫ�Ժ������ܶȝ�������ҕ����һ��늳ؼ��g(sh��)����Ҫ����Ȼ�������y(t��ng)��(f��)�Ϲ̑B(t��i)늽��|(zh��)��CSEs����߾ۺ���Y(ji��)���ȡ��o�C���ψF���Լ�������Ľ���Y(ji��)��(g��u)�ȹ���ȱ�ݣ���K�����x�ӌ�(d��o)��Ե����������Q�@Щ���}���㽭������W(xu��)������ڈFꠏ���Ȼ�������п�ͨ������(d��o)����(ji��)�������ķּ��Y(ji��)��(g��u)�Ы@���`�У��_�l(f��)��һ�N������(ji��)�Y(ji��)��(g��u)��(f��)�Ϲ̑B(t��i)늽��|(zh��)��PHLM-CSE�����ڼ����I(l��ng)���w�S�ͼ����ǘ�(g��u)������Ļ��A(ch��)����Y(ji��)��(g��u)ֱ�ӛQ���˲��ϵ����ܡ��о��Fꠌ��@һ�����w�Ƶ�늳�늽��|(zh��)���O(sh��)Ӌ�У������o늼��z�@һ����Ʒ�Ƃ�ij��ü��g(sh��)�����ۺ��PVDF-HFP���c�o�C���ϣ�LLZTO����(f��)�ϣ����Ƴ����S��{���w�S�Ǽܡ��ṩ�˃�(y��u)���ęCе���g�Ժ��B�m(x��)���x�ӂ���(d��o)·�����S��ͨ�^ԭλ�ԽM�b���w�S���������Եس��eZIF-8�w�����γ����������(ji��)����/�{�������^(q��)���@Щ����(ji��)�����H�@�������늽��|(zh��)Ĥ�ęCе���ȣ�߀�����˸�Ч�Ľ����x�ӂ�ݔͨ����
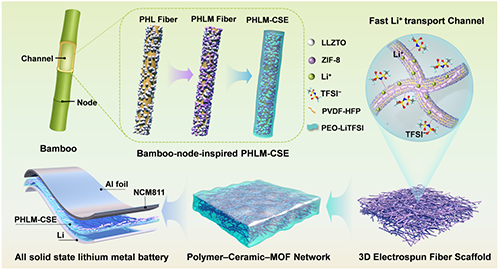
�C��ʾ��D��������(ji��)�Y(ji��)��(g��u)��(f��)�Ϲ̑B(t��i)늽��|(zh��)��PHLM-CSE������ּ��x�ӂ�ݔͨ����ʾ��D��
��D1��ʾ��SEM�Y(ji��)���@ʾ����PVDF-HFP�w�S����⻬�������sLLZTO�������F(xi��n)�{��ͻ�𣨈D1b�����C���o�C�w���ɹ�Ƕ���w�S����(g��u)�ɷ�(w��n)�����������Ǽܡ���(j��ng)ԭλ�ԽM�b��ZIF-8���w����ֲ����w�S���棬�o���@�F�ۣ��D1c�Cd����TEM�Mһ����C���������e�������D1e�Cf�������������CÓ���yԇ������ZIF-8���и߱ȱ���e��1443.5 m2/g���ͷּ��Y(ji��)��(g��u)����������x�ӂ�ݔ���D1h������PEO-LiTFSI����������PHLM-CSEĤ�������ܡ��o�Ѽy���D1g�����@����(y��u)��ֱ�ӝ�����PEOĤ��XRD�cFTIR�Y(ji��)�����D1i�Cj���Mһ���_�J��LLZTO��Ƕ���ZIF-8�ijɹ�ԭλ���L��
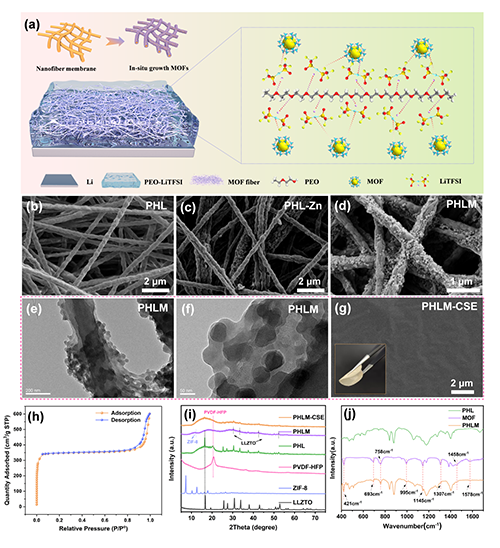
Figure 1. Structural design and morphological analysis of the PHLM-CSE composite solid-state electrolyte. (a) Schematic illustration of the three-dimensional architecture of the PHLM-CSE and the proposed mechanism by which MOF facilitates Li+ transport; (b) SEM image of the PVDF-HFP/LLZTO nanofiber membrane (PHL), scale bar : 2 ��m; (c) SEM image of the PHL-Zn nanofiber membrane after Zn2+ ion adsorption, scale bar: 2 ��m; (d) SEM image of the PHLM nanofiber membrane following in situ MOF growth, scale bar: 1 ��m; (e, f) TEM images of the PHLM nanofiber membrane highlighting internal structure; (g) SEM image of the surface morphology of the PHLM-CSE electrolyte, scale bar: 2 ��m. Inset: photograph image of the as-prepared membrane; (h) Nitrogen adsorption-desorption isotherm of the as-prepared ZIF-8 measured at 77.3 K; (i) XRD patterns of the PHLM-CSE electrolyte, PHLM and PHL membranes, pristine PVDF-HFP, ZIF-8, and LLZTO. Key diffraction peaks for PVDF-HFP, LLZTO, and ZIF-8 are indicated by dotted lines and arrows for clarity; (j) Fourier transform infrared (FTIR) spectra of the PHL, ZIF-8, and PHLM nanofiber membranes.
��D2��ʾ��PHLM-CSE��MOF������F(xi��n)�����͵�Tg��-36.6 ��C�����ߵ�Tm��58.0 ��C��������x���w�����c�᷀(w��n)���ԡ�����я��Ⱥ͗���ģ���@���������߂����(y��u)��֦��������늌W(xu��)���ܷ��棬PHLM-CSEչ�F(xi��n)�����߽�늳���(sh��)�������}���x��������50 ����늌�(d��o)���_5.04 × 10-4 S·cm-1�����߂�5.1 V�Č�늻��W(xu��)���ڣ��C�����ܾ���(y��u)�ڌ��սM��
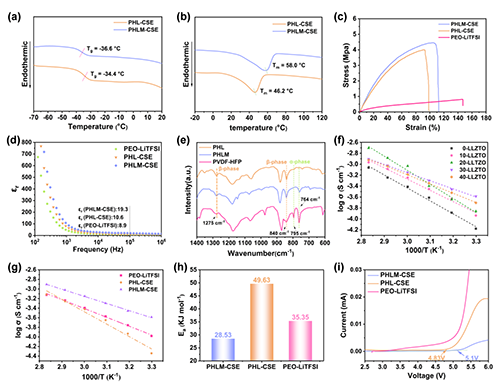
Figure 2. Structural features and electrochemical performance of the PHLM-CSE composite electrolyte membranes. (a) Differential scanning calorimetry (DSC) curves of PHL-CSE and PHLM-CSE electrolytes, highlighting their glass transition temperature (Tg) ; (b) DSC curves of PHL-CSE and PHLM-CSE in the higher-temperature region, illustrating thermal stability; (c) Stress-strain curves of PHL-CSE, PHLM-CSE, and PEO-LiTFSI electrolytes, revealing mechanical strength and flexibility; (d) Frequency-dependent dielectric constant (��r) of PEO-LiTFSI, PHL-CSE, and PHLM-CSE electrolytes; (e) FTIR spectra of PHL, PHLM and pristine PVDF-HFP membranes, demonstrating polymer phase characteristics and structural features; (f) Arrhenius plots of ionic conductivity (��) versus temperature for PHLM-CSE electrolytes with varying LLZO contents; (g) Comparative ionic conductivity-temperature profiles of PEO-LiTFSI, PHL-CSE, and PHLM-CSE electrolytes; (h) Bar graph comparing the lithium-ion migration activation energy (Ea) across different electrolytes; (i) Linear sweep voltammetry (LSV) curves evaluating the electrochemical stability window of the investigated membranes.
��D3��ʾ��PHLM-CSE��50 ��C�±��F(xi��n)�����ٷ�(w��n)���ĺ㉺�O������������x���w�Ɣ�(sh��)���_0.64���h����PHL-CSE��0.26����PEO-LiTFSI��0.14�����D3a,b����EIS�Y(ji��)���@ʾ������迹�������L�ڷ�(w��n)�����D3c�������Q����ܶ��_14.2 ��A cm-2���D3d��������0.8 mA cm-2�±��ַ�(w��n)��늉�ƽ�_���D3e�����f����߂䃞(y��u)���Ľ�������W(xu��)���ߵ��R������ܶȣ���Ч�����֦�����L������ӽY(ji��)��(g��u)�Ͽ���PVDF-HFP�ĵ�HOMO�ܼ��x���wϵ���ÿ������ԣ���ZIF-8�cLiTFSI�ĵ�LUMO�ܼ������ڃ�(y��u)���γɷ�(w��n)��SEI���D3f������Ǿ����o늄ݷֲ����D3g���c�^�߽Y(ji��)���ܣ��D3h���Mһ���������淀(w��n)�����cLi+�x�����w��Ч�ʡ�
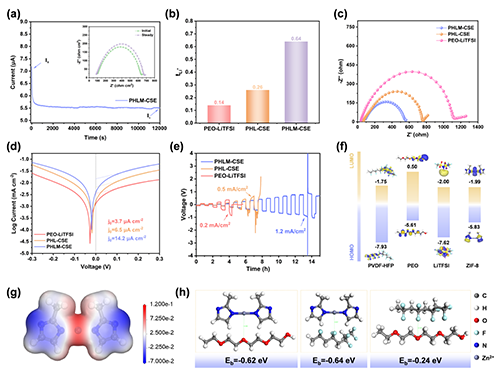
Figure 3. Electrochemical properties and interfacial stability analysis of the PHLM-CSE composite solid-state electrolyte. (a) Galvanostatic charge-discharge profiles of the PHLM-CSE-based cell, with the inset displaying the corresponding electrochemical impedance spectroscopy (EIS) data; (b) Comparison of lithium-ion transference numbers (tLi+) for PEO-LiTFSI, PHL-CSE, and PHLM-CSE electrolytes; (c) Electrochemical impedance spectra (EIS) of symmetric Li|PEO-LiTFSI|Li, Li|PHL-CSE|Li, and Li|PHLM-CSE|Li cells, reflecting interfacial resistance; (d) Linear sweep voltammetry (LSV) curves used to evaluate the electrochemical stability windows of different electrolytes; (e) Critical current density (CCD) measurements of Li|Li symmetric cells with PEO-LiTFSI, PHL-CSE, and PHLM-CSE electrolytes; (f) Molecular structures and corresponding HOMO-LUMO energy level diagrams of PVDF-HFP, PEO, LiTFSI, and ZIF-8; (g) Electrostatic potential distribution map of ZIF-8, indicating charge localization characteristics; (h) Binding energies (Eb) and optimized configurations of ZIF-8 interacting with PEO, PVDF-HFP, and their binary complex.
��D4��ʾ��PHLM-CSE��TFSI-�Խ��x�B(t��i)������83.3%�������@����PHL-CSE��71.2%����PEO-LiTFSI��20.4%�����D4a��������MOF������Li+�CTFSI-���á������}���xЧ�ʡ�MDģ�M�@ʾPHLM-CSE��Li+ MSD����б����ߣ��D4c����RDF�cCN�Y(ji��)�����D4d,e���Mһ���C�����Li+�ṩ����λ�h(hu��n)��������Uɢͨ�����Ķ��@�������w����������Li|Li���Q늳��У�PHLM-CSEѭ�h(hu��n)��(w��n)���Գ��^2700Ȧ���D4f�����h��(y��u)��PHL-CSE��1220Ȧ����PEO-LiTFSI��239Ȧ����XPS�cEDS�������D4h��S22������PHLM-CSE�������ɾ����LiF����(d��o)SEIĤ������TFSI-�ֽⲢ�����x���w��Ч�ʡ���ò�������D4h�Cj����ԭλ�@�^�죨�D4k�����@ʾ�����ƽ�����o֦���γɣ��Mһ����C����Խ�Ľ��淀(w��n)�����c��֦��������
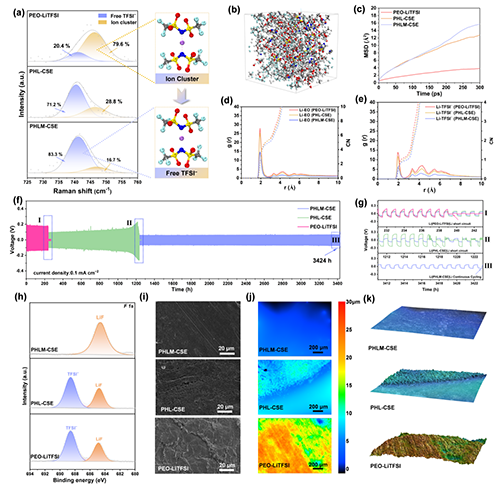
Figure 4. Investigation of lithium-ion transport behavior and interfacial stability in PHLM-CSE composite solid-state electrolytes. (a) Raman spectra of PEO-LiTFSI, PHL-CSE, and PHLM-CSE composite electrolytes, highlighting variations in ion coordination environments; (b) Snapshot from a molecular dynamics (MD) simulation of the PHLM-CSE model system; (c) Mean squared displacement (MSD) curves of Li+ in PEO-LiTFSI, PHL-CSE, and PHLM-CSE electrolytes, reflecting ion mobility; (d) Radial distribution functions (RDFs) and coordination environments between Li+ and TFSI- in various electrolytes; (e) RDFs and coordination environments between Li+ and ether oxygen (�CO�C) atoms in the EO segments; (f) Galvanostatic cycling performance of Li|Li symmetric cells with PHLM-CSE, PHL-CSE, and PEO-LiTFSI electrolytes at 0.1 mA cm-2. (g) enlarged voltage profiles near the short-circuit or failure points in Li|PEO-LiTFSI|Li, Li|PHLM-CSE|Li, and Li|PHL-CSE|Li cell; (h) F 1s core-level XPS spectra of Li anodes after 100 cycles with different electrolytes; (i) SEM images of Li metal surfaces after cycling; (j) Two-dimensional laser confocal microscopy imagesshowing cycled surface morphology; (k) Three-dimensional laser confocal microscopy reconstructions of Li metal surfaces following long-term cycling.
��D5��ʾ��LiFePO?|PHLM-CSE|Li늳���0.2�C2 C�±��ָ߱�������144.2��83.3 mAh g-1�������ʻ֏�(f��)���ã��D5a,b����ѭ�h(hu��n)200Ȧ�������������_97.2%���D5c������1 C�¿ɷ�(w��n)���\��600Ȧ���ϡ��Mһ����NCM811�wϵ�У�PHLM-CSE늳���1 C��ѭ�h(hu��n)800Ȧ���Ա���95.2%�������D5e,f�����@����(y��u)��PHL-CSE��PEO�wϵ���ұ��F(xi��n)�������m��(y��ng)�ԣ�20�C60 ��C�����@�N��(y��u)��Դ��MOF���M�}���x�cLi+�w�ƣ��Լ�LLZTO�������淀(w��n)���Ժ����W(xu��)���ȡ��ڑ�(y��ng)�÷��棬����PHLM-CSE������ܛ��늳��ڏ��ۡ��ۯB�����̼����汩¶�����ܷ�(w��n)���(q��)��LED���D5g�������ڼ�������늂������c���ܽK���б��F(xi��n)����(w��n)����늣��D5h,i��������0.2 C�������s170 mAh g-1�����Ч��>99%����߸������ܶȡ���ȫ�Ժ����ԣ�չʾ���ڿɴ�����Դϵ�y(t��ng)�еďV韑�(y��ng)��ǰ����
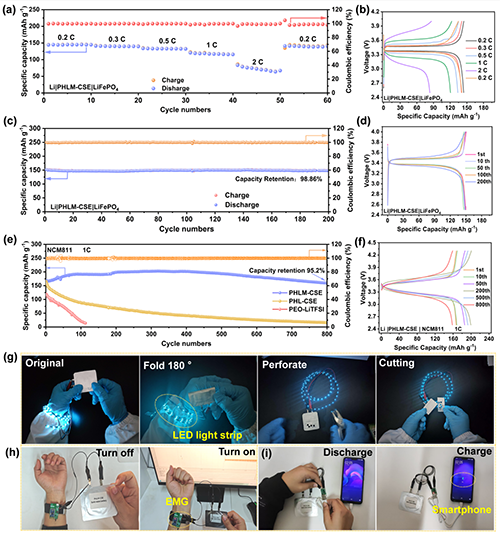
Figure 5. Electrochemical performance and flexible applications of PHLM-CSE-based solid-state lithium metal batteries. (a) Rate capability and (b) corresponding charge�Cdischarge profiles of LiFePO4|PHLM-CSE|Li full cells at various current densities. (c) Long-term cycling performance at 0.2 C and (d) corresponding voltage profiles. (e) Cycling stability of NCM811|PHLM-CSE|Li, NCM811|PHL-CSE|Li, and NCM811|PEO-LiTFSI|Li full cells at 1 C, and (f) their voltage profiles. (g) Optical images showing stable operation of a pouch cell under flat, folded, cut, and punctured conditions. (h,i) Demonstration of the pouch cell powering an electromyography (EMG) sensor and a smartphone, respectively.
��֮�����о�����(ji��)�Y(ji��)��(g��u)���l(f��)����(g��u)���˷�����(f��)�Ϲ̑B(t��i)늽��|(zh��)��PHLM-CSE����ͨ�^�o늼��z�cԭλZIF-8�M�b�γ��B�m(x��)�x��ͨ���ͷ�(w��n)�̾W(w��ng)�j(lu��)���@���������x�ӂ�ݔ�c���W(xu��)���ȡ�PHLM-CSE��50 ��C�¾߂���x�ӌ�(d��o)��ʣ�5.0 × 10-4 S·cm-1�������w�Ɣ�(sh��)��0.64������늻��W(xu��)���ڣ�5.1 V���̓�(y��u)�����W(xu��)���ܡ�늳yԇ�@ʾ�����L���������Q늳�>3400 h�����߷�(w��n)���ԣ�NCM811ȫ늳�800Ȧ������95.2%�������ԑ�(y��ng)�Ý������錍�F(xi��n)�����ܡ�������(w��n)���Ĺ̑B(t��i)䇽���늳��ṩ����˼·��
�ڴˣ����x�㽭ʡ��Ȼ�ƌW(xu��)�����Ŀ��LY21E030023�����㽭������W(xu��)���݄�(chu��ng)���о�Ժ�����Ŀ��SYY2024C000008����֧�֣�
ͨӍ���ߺ��飺���㣬�У���ʿ�����ڣ���ʿ����(d��o)�����㽭������W(xu��)�����ƌW(xu��)�c���̌W(xu��)Ժ�����H�z�I�W(xu��)Ժ����Ժ�L����Ҫ���·�ˮ���|(zh��)Ⱦ���¼��g(sh��)������������ܼ���Ʒ�о����Ե�һ����ͨӍ������Advanced Functional Materials, Energy Storage Materials,Advanced Fiber Materials, Nano Letters, Nano Energy�ȿ����ϰl(f��)��SCIՓ��70��ƪ���ڙ�(qu��n)���D(zhu��n)�����Ұl(f��)������30��헡��@�Ç��Ҽ��̌W(xu��)�ɹ����Ȫ����㽭ʡ�̌W(xu��)�ɹ��صȪ���1헣����֫@���Ї��������I(y��)(li��n)�ϕ��̌W(xu��)�ɹ�һ���������Ȫ����㽭ʡ��Ȼ�ƌW(xu��)�����Ȫ����Ї��̘I(y��)(li��n)�ϕ��Ƽ��M�������Ȫ���1헡�
ԭ��朽ӣ�https://doi.org/10.1002/adfm.202514738
- ���t(y��)��һ��Ժʢ�������/�A�ƴ��R������ AFM����(g��u)���˷������ˮ���z�NƬ-�ڼ���p���ί�����ȡ�����Mչ 2026-03-02
- ����������·���ڈF� AFM���ġ����顱���ֵ��IС������{���w�SĤ - ���F(xi��n)�{��Һ��ͨ�����x 2026-02-25
- �|�A��W(xu��)�������о��T/ʷ��ꖽ��ڈF� AFM�����������w���b��pH푑�(y��ng)�͘�����Ӽ{�����z��������ͨ�^�ؾ��̶�N��������ί����ٰ� 2026-02-20
- ���ڴ�W(xu��)���� AFM�����ڸ߷�(w��n)���Թ̑B(t��i)䇽���늳ص��x�ӌ�(d��o)늷��ӽ�֦�ɳ��m(x��)�w�S�ػ��ʹ��w��(f��)��늽��|(zh��) 2024-05-10
- �������i�w����������Sci. China Chem.��Highlight��ͨ�^�մ�/�ۺ�������Ч��ݔ��x�ӵď�(f��)��늽��|(zh��) 2023-03-31
- ���ϴ�W(xu��)���������ڈF� Small��MOF���w�S�W(w��ng)�j(lu��)�����ۺ����(f��)��늽��|(zh��)���F(xi��n)������ȫ�̑B(t��i)�c����늳� 2023-01-26
- ������������� Nano Energy�����ڮ��|(zh��)���湤��Janus�Y(ji��)��(g��u)��(f��)�ϼ{���w�SĤ���F(xi��n)������ȫ�̑B(t��i)䇽���늳� 2025-05-20
�\���P(gu��n)ע�߷��ӿƼ�

- 2026����ߌ��Ї�����Ұl(f��)չ...
- �����I(l��ng)Ʊح300+���h��I(y��)�R��...
- �P���Ж|�����L���Ѓx�����{...
- �f����Ŀ �ذ��_�֣�2025�C...
- ICIE���H(�V��)Ϳ�Ϲ��I(y��)չ�[...
- �_չ�ڼ����ۄ�������朄�δ...
- 9������Ҋ���£�AI�(q��)��+���a(ch��n)...
- �P(gu��n)���e�k�ڶ�ʮ�����Ї����H...
- �R�ۼ�˃x�� ����ȫ���̙C ...
- CPHI����չ����҃x���c�O(sh��)��...
- ���ܹ��A��朆���������22��...
- ������/���f/�Y�� JACS���l(f��)...
- ��ɽ��W(xu��)��־�x�����w�����...
- ���������������ֈF� Angew...
- �Ϻ�����/�����������...
- ���t(y��)��һ��Ժʢ�������/�A...
- �пƴ���Ƙ�������������/...
- �����������/������������...
- �㹤��������/�~��Ҋ�n�}�M ...
- ��������-��־�F� AFM �C��...
- �|�ϴ�W(xu��)��������ڈF� Nat...
- ���ݴ�W(xu��)Ҧ����/κ��/����Ӱ...